 You don't need to calculate, but calculating is always just safe to do. So I hope that you guys see how, if it fits, it's bonding perfect. This is another way that you can fulfill your bonding preference by having a double bond and having to lone pairs, it still counts is to Bonds and two lone pairs. And it has six electrons total in terms of the valence 66 sticks and dots. Okay, Um so basically, for this one on the oxygen has six electrons total. Okay, then finally, there was a type of just so you know, So notice that in your page the oxygen did not have those lone pairs. So now you do that for carbon, but its carbon foot fulfilling its bonding preference. So then I would get a formal charge of zero, See how easy that is. So how maney is that All together? That's six. It's six minus the number of sticks and dots. So we said that the formal charge equals the group number. 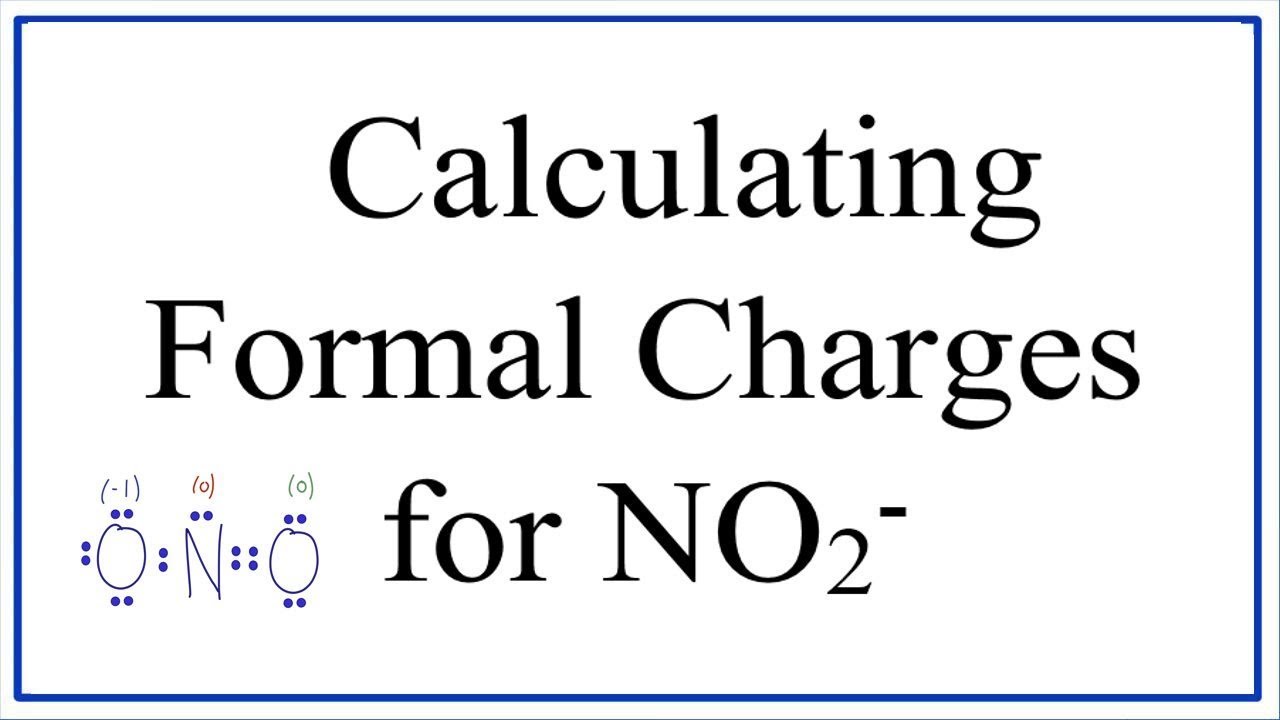
Okay, but now I want to show you mathematically how it works out to. Okay, So first of all, does this oxygen fit my bonding preference According Thio Bond represent, Yes, it does. Let's do this with this oxygen right here. So as long as you're bonding, preference agrees with what you see, that's going to be zero. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators. Do you see that? But it also fits with my bonding preference. My equation says that formal charge equals group number, which is one minus sticks and thoughts, which is one. So it's gonna be one according to the equation. Remember that Bonding preferences say that hydrogen wants toe. So let's start off with the hydrogen even though we already know that this fits the bonding preference. What that means is I'm gonna be looking at group numbers and I'm gonna be looking at sticks and thoughts. Let's go ahead and do this example where I wanna look at each atom and I want to count the formal charges for all of them. Using the structure of SCl6 S C l 6, calculate the formal charge of the central sulfur atom. There's usually not that many formal charges on the molecule. The formal charge on the central phosphorus atom in the following structure is: 26. In general you want: the fewest number of formal charges possible, i.e. No Lewis structure is complete without the formal charges. You're just looking at each Adam and saying, Does this have a formal charge? Does this have a formal charge? The net charges the collection of all of those sums together. The formal charge is calculated by: (group number of atom) - (½ number of bonding electrons) - (number of lone pair electrons), i.e. You see it a little bit confused over like, Oh, does the formal charge go on the whole thing? Or is it just one, Adam? No. Now, this is an important point because I remember when I was an undergrad. The net charge is the term that we give for the sum of all the formal charges. So you take your group number, then you just subtract the sticks and the dots and you're good. A lot of times, you'll just be able to do this on your fingers. And then you subtract the valence electrons, which is just the sticks and the dots. So all you do to calculate formal charge is you take the group number, whatever that is, that could be Group four, Group five, whatever. So remember that the group number is how maney it wants the valence electrons, the sticks and the dots are the it actually has. Basically, a formal charges assigned whenever there's a difference between the number of Valence electrons and Adam wants toe have and the number of valence electrons it actually has. So let's go ahead and just jump right into it. An online formal charge calculator is exclusively designed to calculate formal charge of an atom. 
And formal charges are just based on the entire idea of bonding preferences. Also, it places the least electronegative atom in the center, and the negative charge on the more electronegative element (Guideline 4).So now that we understand bonding preferences so well, I want to move to a really related topic called formal charges. However, the first arrangement of atoms is preferred because it has the lowest number of atoms with nonzero formal charges (Guideline 2). Note that the sum of the formal charges in each case is equal to the charge of the ion (–1). Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
